 
The particles in the metal are held together by strong metallic bonds.Delocalized electrons exist between the particles in the metal.Which structural property of metals is responsible for their hardness? Tend to have a relatively low electron affinity and ionization energy because they have such a large atomic radius.Įxample 1: Understanding Why Metals Are Generally Stiff and Hard Substances Valence shell that is less than half full, and they have an atomic radius that is larger than nonmetals of the same period. Metals are known to have a relatively low number of valence electrons and a relatively large atomic radius. That contain steel skeletons or some other types of metal support structures. There also happens to be an unimaginably high number of large buildings and monuments Vehicles like cargo ships and even airplanes. Hard metals are regularly used to make cars, and they are also used to make larger transport Most metals are strong, and this makes them suitable for constructing vehicles and for making the structural support components Metallic bonding is the strong electrostatic attraction that exists between positively charged metal cations and delocalized Mercury is used to make thermometersīecause it has a low melting point and it is a liquid at room temperature. Mercury has the lowest melting point of all the metallic elements. The filaments of most light bulbs, specifically because it can glow white-hot without melting. Tungsten metal has the highest melting point of all the metallic elements. Strongest metallic bonds of any pure metal element. Tungsten atoms are held together with some of the It takes a lot of heat or mechanical energy to break them apart. There are strong electrostatic attraction forces between the metal cations and the delocalizedĮlectrons. Metals are known to be hard and to have high melting points and boiling points because they are made up of metal cations inĪ sea of delocalized electrons. Very different physical and chemical properties, and they are each very well suited for a set of specific industrial and Metals are not more useful than nonmetals, and nonmetals are not necessarily any more useful than metals. 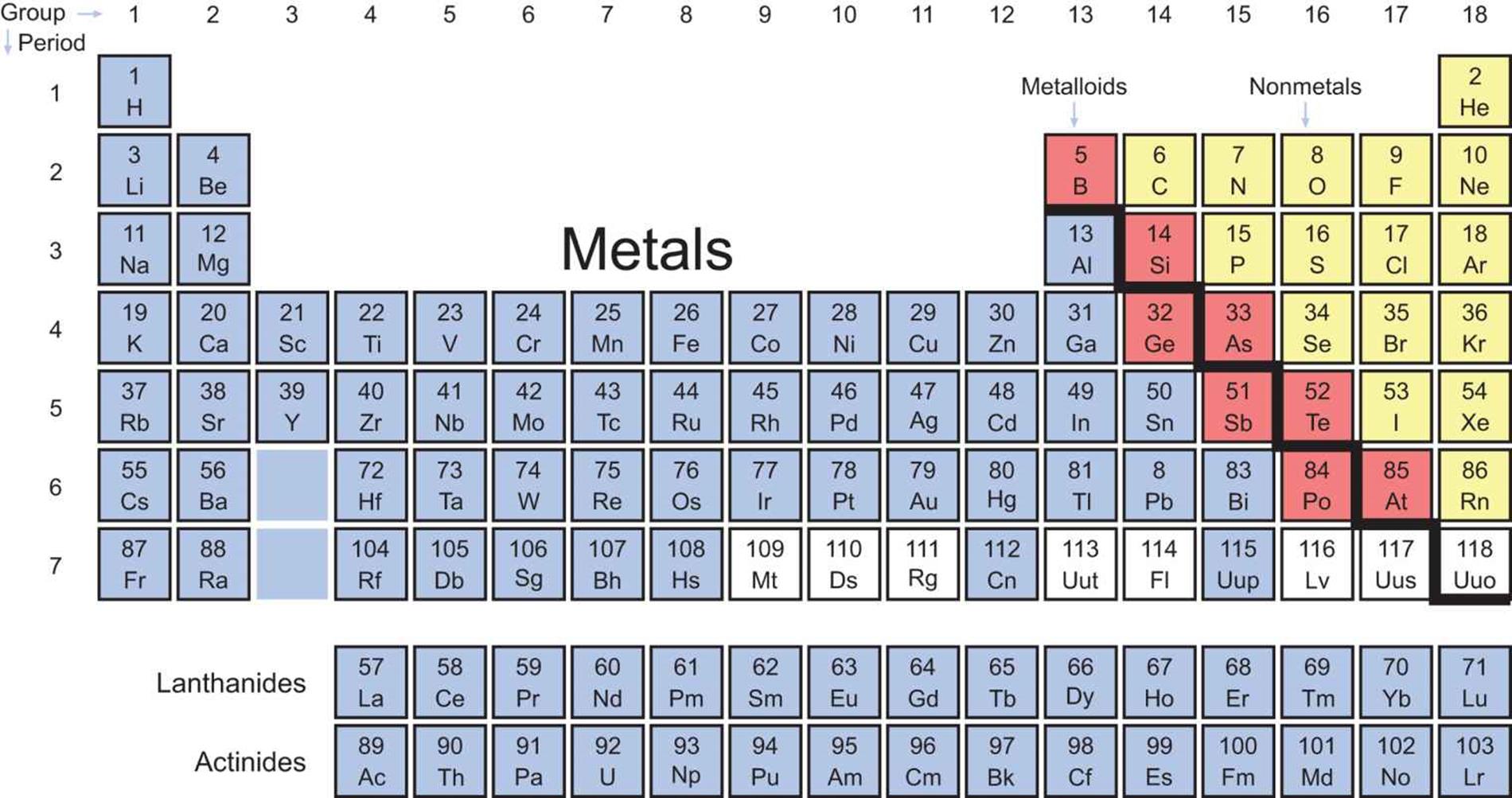 
Nonmetal atoms usually form much smaller covalently bonded molecules. Positively charged ions in a sea of delocalized electrons. Metal atoms usually form giant three-dimensional structures that are made up of billions of Metal and nonmetal elements have very different chemical and physical properties because they are made up of atoms that areīonded in very different ways. The white cells represent chemical elements with unknown chemical properties. The metalloid elements are represented with a yellow color, and they can be found in between the left- and right-hand The nonmetal elements are represented with the color blue, and they can be found on the right-hand side of the periodic The metal elements are represented with the color red, and they can be found on the left-hand side of the periodic The following figure uses red and blue colors to show how the periodic table transitions from metal elements to nonmetalĮlements. His classifications were based on the physical He classedĮlements as metals or nonmetals more than one-hundred years ago. Jöns Jacob Berzelius was a 19th century scientist considered to among the founders of modern chemistry. The periodic table has been divided into separate metal and nonmetal sections for a relatively long time now. It can be explained with relatively simple theories about the exchange of valence electrons and intermolecular bonding Phenomenon is fascinating not only because it is maintained for almost all rows of the periodic table, but also because Metalloids and then to nonmetals as we move from the left-hand side to the right-hand side of the periodic table. The periodic table of elements has many interesting trends, and one of the most obvious is the transition of metals to In this explainer, we will learn how to describe, compare, and explain the physical and chemical properties of metals, 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
